Scientists at UNIST have developed a new low-cost catalyst synthesis method that can efficiently decompose water into oxygen and hydrogen using waste-yeast biomass. This system has a high return, and in this way, is expected to additionally decrease the expense of creating hydrogen from the decomposition of water.
Besides, by covering the yeast-based support with ruthenium (Ru) and iron (Fe)- based materials, they built up another catalyst material that displays astounding execution in both hydrogen and oxygen generation.
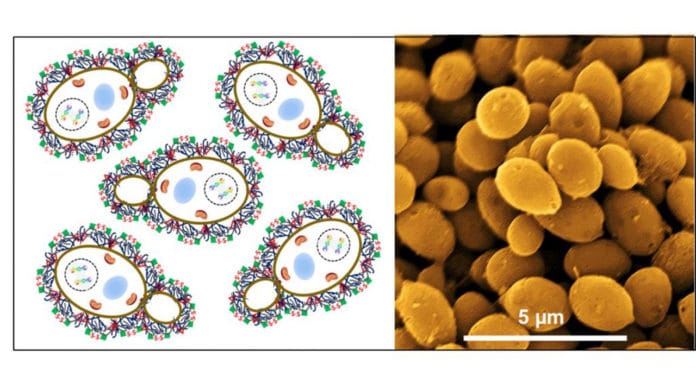
Scientists focused on the squander yeast biomass, as catalyst material that will improve both oxygen and hydrogen generation, while supplanting the noble metal catalysts, for example, Pt or Ir. As yeast is a living organism, it is wealthy in substances, for example, carbon (C), phosphorus (P), sulfur (S), and nitrogen (N).
Such materials can have an increase in electrical conductivity, yet additionally, practical gatherings that can hold different materials to assist fix metal particles. This feature can make a suitable catalyst, eventually.
In the study, the research team has created two catalysts that promote the generation of both hydrogen and oxygen, using waste yeast as catalyst support. They reported hydrogen and oxygen production in 1 M potassium hydroxide using ruthenium single atoms (RuSAs) along with Ru nanoparticles (RuNPs) embedded in MHC (RuSAs + RuNPs@MHC) as a cathode and magnetite (Fe3O4) supported on MHC (Fe3O4@MHC) as an anode.
The RuSAs + RuNPs@MHC catalyst outperforms the state-of-the-art commercial platinum on carbon catalyst for hydrogen evolution reaction in terms of overpotential, exchange current density, Tafel slope, and durability. Furthermore, compared with industrially adopted catalysts (that is, iridium oxide), the Fe3O4@MHC catalyst displays outstanding oxygen evolution reaction activity.
Scientists noted, “In particular, for whole water splitting, it requires a solar voltage of 1.74 V to drive ~ 30 mA, along with remarkable long-term stability in the presence (12 h) and absence (58 h) of outdoor-sunlight exposure, as a promising strategy towards a sustainable energy development.”
Distinguished Professor Kwang S. Kim (National Honor Scientist of Korea) in the School of Natural Sciences at UNIST said, “Yeast biomass-derived materials can help develop efficient, eco-friendly and economic catalysts to improve the sustainability of hydrogen production. Compared to coal and oil, waste yeast is eco-friendly, inexpensive, and readily available biomass, and the results of the study suggest a new application of waste yeast biomass.”
This study has been participated by Research Professor Jitendra N. Tiwari (School of Natural Sciences, UNIST), as a co-corresponding author. The National Honor Scientist Program has supported it via the National Research Foundation of Korea (NRF) and the Ministry of Science and ICT (MSIT).
Journal Reference:
- Multi-heteroatom-doped carbon from waste-yeast biomass for sustained water splitting. DOI: 10.1038/s41893-020-0509-6