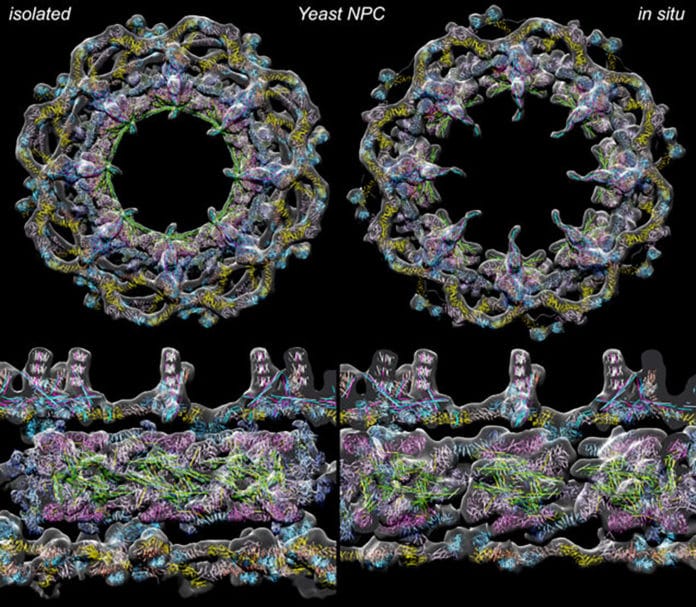
The nuclear pore complex (NPC) is a crucial regulator of molecular traffic between the cytoplasm and the nucleus. It transports molecules into and out of the nucleus.
Defects in NPC are associated with many diseases such as viral infections, cancers, and certain neurodegenerative diseases. In the latest study, scientists at the Boston University School of Medicine have created a comprehensive model of the yeast NPC.
Scientists used rapid plunge freezing and cryo-EM (electron microscopy) with computational methods for this task. The work provides molecular models for two configurations: one that is easier to study in isolated samples to give a more detailed overview of a radially-compact form and a second expanded form in the living yeast cell, albeit this “in situ” form is currently visualized with a lower level of detail.
Corresponding author Christopher W. Akey, Ph.D., professor of physiology and biophysics, said, “This research significantly extends our understanding of the architecture of the NPC from brewers’ yeast, a model organism that is used to study the biology of cells that contain a nucleus and thus provides new insights on multiple levels into the functions of this transport machine.”
The model offers a detailed understanding of how these large mega-channels assemble and how they can flex and adapt to transport changes by expanding their central passageway.
They said, “Moreover, we have observed multiple types of NPC in the same cell for the first time, which reflects the lego-like ability of this assembly to use interchangeable parts to modify its architecture on the nuclear side. This adaptability may play a role in tailoring the functions of these machines for different local environments at the periphery of the nucleus.”
The study could help scientists reveal how viruses may usurp critical pathways to infect cells and alter their physiology to cause disease.
Journal Reference:
- Christopher W. Akey et al. Comprehensive structure and functional adaptations of the yeast nuclear pore complex. DOI: 10.1016/j.cell.2021.12.015