Inspired by chameleon skin that changes its skin color in response to its environment, engineers at Iowa State University have developed a new way for liquid metal – and potentially solid metal – to change its surface structure in response to heat.
They have developed chameleon metal that could potentially inspire the design of ‘smart’ alloy systems that evolve the surface patterns and their composition with temperature (or analogous stimuli) for applications ranging from sensing to catalysis.
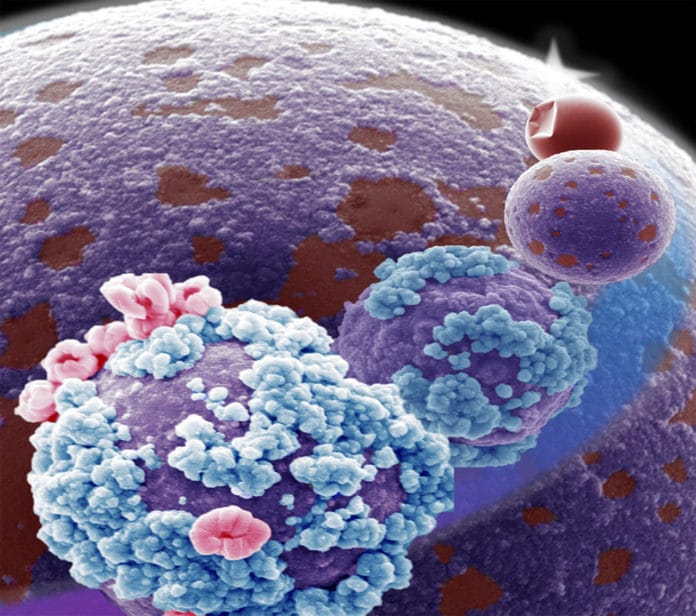
Treating particles of liquid metal alloys with heat causes them to roughen their surfaces with tiny spheres or nanowires. To do so, scientists started with an alloy of gallium, indium, and tin synthesized into particles covered with a smooth oxide shell that has been chemically stabilized. As the particles are warmed, the surface thickens and stiffens and starts behaving like solid.
In the long run, the surface breaks, permitting the liquid metal inside to rise to the top. The most reactive, gallium, breaks through first. More heat carries indium to the surface. Also, the most noteworthy temperature – around 1,600 degrees Fahrenheit – brings out florets of tin.
This movement from the under-layer to the surface enables a liquid metal particle to invert its composition under thermal stimuli constantly.
Martin Thuo, an Iowa State assistant professor of materials science and engineering, said, “The particles are responding to a certain level of heat and releasing a specific element based on temperature, just as a chameleon responds to the color of its environment. That’s why we say they’re chameleon metals – but responding to heat, not to color as the reptile does.”
“The metal particles are responding to a very controlled environment – the researchers carefully control time, temperature, and oxygen levels.”
This allowed scientists to predict and program the exact surface texture of the particles.
Martin said, “the technology could be used to fine-tune a metal’s performance as a catalyst or its ability to absorb compounds.”
Thuo said, “This is not unique to these materials. This is a behavior of metals in general. Other metals subject to the same treatment should do this. This is a universal property of metals.”
The paper is presented in the issue of the journal Angewandte Chemie.