Biology is swaddled in lipids: fats, oils, and even waxes encompass cells and their organelles, intercede the stream of immense biological data systems, ensure delicate tissues, and store fundamental energy over different living beings.
Lipids have customarily been among the hardest biomolecules to examine in view of the assorted variety of their sub-atomic structures, which are not dictated by the very much characterized building blocks and simple rules that oversee DNA, RNA, and proteins. Also, this diversity means, unlike building and investigating genome and transcriptome databases, lipids require more redid systematic techniques.
Along these lines, it is extremely hard to think about either the physiological function of a greater part of lipid species or the way they are so correctly directed in cells. Be that as it may, while lipidomics innovations are advancing, making an interpretation of their discoveries into medical applications and bringing them into clinical research centers is as yet a significant test.
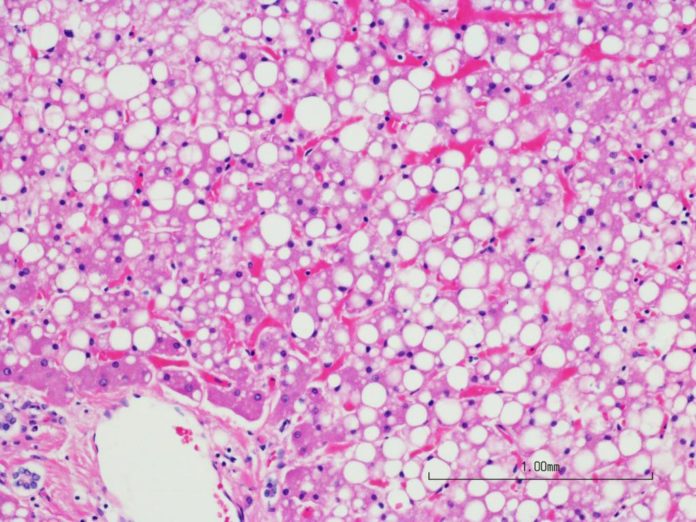
This is the challenge that the team of Johan Auwerx at EPFL, in collaboration with Dave Pagliarini’s group at the University of Wisconsin-Madison took on by measuring almost 150 lipid species in the blood and liver of mice. Scientists have carried out one of the most extensive lipidomics studies to date, connecting almost 150 different lipid species to their respective genetic regulators, revealing signatures of metabolic health and disease.
Scientists used systems genetics approaches to merge the lipidomics information with other “omics” datasets (phenomics, proteomics, transcriptomics) from this populace of mice (BXD). The approach distinguished plasma and blood lipid species from various lipid classes as signatures of healthy or undesirable metabolic states.
They demonstrated seven plasma triglyceride species as signatures of healthy or fatty liver and non-alcoholic fatty liver disease (NAFLD). Their perception was approved in an autonomous dietary and therapeutic model of NAFLD in mice and in plasma from patients with NAFLD.
Johan Auwerx at EPFL said, “This finding fuels optimism that lipid species could serve as signatures or biomarkers that will replace the invasive tissue biopsies currently used to diagnose diseases such as NAFLD – simply by measuring specific lipid species in the blood.”
In a second study, scientists identified signatures of healthy or fatty liver a subset of the cardiolipin lipids, which are the essential phospholipids in the inner membrane of mitochondria.
In both studies, scientists pinpoint a few genetic areas that may control the creation of lipid species. By looking at the hereditary information from the BXD mouse populace to information from genome-wide association investigations of lipid-related disorders in people, they could distinguish normal qualities amongst mice and people that direct lipids.
Auwerx said, “Analyzing lipids and finding their physiological role may never be as straightforward as studying nucleic acids or proteins. But these companion studies provide a foundation for understanding the genetic regulation and physiological significance of lipid species, while yet again demonstrating the potential of Big Data mining to address biological and clinical questions.”
Their studies are published in the journal Cell Systems.