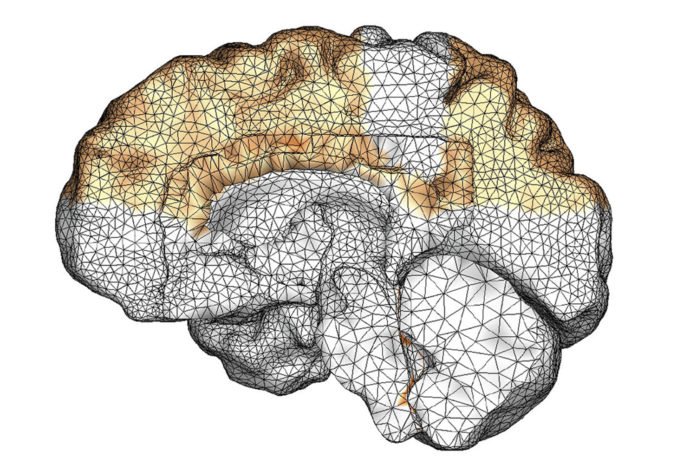
Out of the blue, researchers have built up a computer simulation of how clusters of damaged proteins in neurodegenerative illnesses like Alzheimer’s spread through the cerebrum, a great part of the time in stealth mode, over as long as 30 years.
The simulations focus on Alzheimer’s, Parkinson’s and amyotrophic lateral sclerosis (ALS, or Lou Gehrig’s disease), but the researchers believe their technique is general enough to work for other brain disorders that involve misshapen proteins, including chronic traumatic encephalopathy.
Stanford mechanical engineer Ellen Kuhl said, “We hope the ability to model neurodegenerative disorders will inspire better diagnostic tests and, ultimately, treatments to slow down their effects.”
Scientists were aware that each of the three diseases they were studying produced hallmark clumps of defective, misfolded proteins that build up in the brain.
To perceive how those toxic clumps spread over the long run, scientists took a gander at brain slices taken from individuals who kicked the bucket in the wake of contracting one of the three diseases. Earlier specialists had recolored those brain slices to uncover the presence of the different proteins of interest.
At the point when scientists put the subsequent information into a computer, they additionally did the scientific demonstrating to mimic how the pattern of damaged proteins spreads from the generally inadequate clumps in individuals who were in the malady to substantially more across the board clumping in individuals with advanced illness – a procedure that can take up to 30 years.
Kuhl said, “Imagine a domino effect. What our model does is connect the dots between the static data points, mathematically, to show disease progression in unprecedented detail.”
In the case of Alzheimer’s disease, the scientists modeled the progression of two misfolding proteins – known as tau and amyloid beta – both of which change shape and form toxic clumps in the brains of people with the disease. Prior researchers had stained brain slices for both proteins and, with the new model, Kuhl’s team was able to create two simulations showing the different way that each of these variants of that disease spread.
Neuroscientists do not know precisely how one clump of defective proteins affects its neighbors to spread the misfolding, although Kuhl said there are three prevailing theories. The virtue of the model, she said, is that it predicts the path of the disease regardless of which theory is correct.
Kuhl now plans to work with neuroscientists to better understand the mechanisms of how the proteins misfold. These insights would improve their model and perhaps lead to better ways of diagnosing the disease while it is still in stealth mode.
Kuhl said, “The real challenge is that cell death from toxic proteins occurs years, if not decades, before the first symptoms begin to show.”
“Given the aging of the population, by mid-century 135 million people worldwide will have some form of dementia. We have to find new ways to spur research toward diagnostics and interventions, and computer modeling can play a key role in identifying new therapeutic targets.”
The study is published in the journal Physical Review Letters.